Empirical Formula of Magnesium Oxide Lab Conclusion
Oxygen 15999 gmole 1. 3 762 words Journal Article Critique of an Empirical Analysis of Trends in Psychology Pages.

Empirical Formula Lab Conclusion Magnesium Oxide Youtube
Magnesium Oxygen 008g 005g Magnesium Oxide 013g It is found that there is no difference in mass between the left side of 013g of Magnesium Oxygen to the right side of 013g of Magnesium Oxide in the equation considering the possibility of experimental errors which represents a positive outcome as an theoretical equation is established and.

. Experimental empirical formula of magnesium oxide. The Empirical Formula of Magnesium Oxide The mass of Mg the mass of O2mass of MgxOx. In this lab you will experimentally determine the empirical formula of magnesium oxide the compound formed when magnesium metal reacts.
Use the following molar masses. Up to 24 cash back The empirical formula of magnesium oxide MgxOy can be written based on the lowest whole-number ratio between the moles of Mg used and moles of O consumed. The Empirical Formula of Magnesium Oxide Empirical Formula Lab Conclusion -- Magnesium Oxide.
Lab Assignment 1 Pages. Moles of oxygen 6. B The mass of oxygen that combined with the magnesium was 02 g.
Mass of Mg 2. The Empirical Formula of Magnesium Oxide Empirical Formula Lab Conclusion --Magnesium Oxide Finding the Empirical Formula for Magnesium Oxide part 2wmvEMPIRICAL FORMULA OF MAGNESIUM OXIDE -. Experimental Determination of Empirical Formula of Magnesium Oxide - DATA COLLECTIONEmpirical Formula of Magnesium Oxide Post-Lab Lab.
𝑴 𝒔𝑶𝟐 𝑴 𝑶 𝒔 The lowest whole number ratio of moles of magnesium atoms to moles of oxygen atoms present in magnesium oxide will give the empirical formula of magnesium oxide. Thus the empirical formula of hydrogen peroxide is HO. Mass of magnesium oxide.
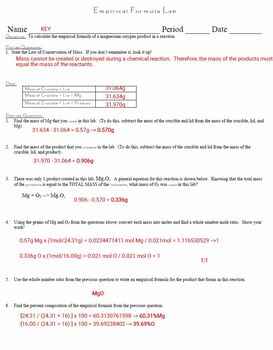
To form solid magnesium oxide Mg x O y. 0011875mol 1 Formula MgO MgO Experimental percent oxygen in Magnesium Oxide Mass of MgO 0190g 0190g0500g x 100 38 Experimental percent magnesium in Magnesium Oxide Mass of magnesium 0310g 0310g0500g x 100 62 Theoretical percent oxygen in Magnesium Oxide Formula. Finding the Formula of Magnesium Oxide Purpose A Lab report by Benjamin Rositsan Lab Partner Madeline Renshaw The purpose of this lab experiment is to experimentally determine the empirical formula of magnesium oxide.
Verify this for yourself as an exercise. For some compounds the empirical formula is also the molecular formula NaCl or H 2O. 3 885 words Projectile Motion Lab Report.
We used the measured mass of magnesium metal and magnesium oxide to determine the empirical formula of the oxide product. Mass of oxygen that reacted with Mg. Give the empirical formula and correct stock system name for the compounds below from the following data and show work for partial credit.
The empirical formula of this molecular compound is C 2 H 5 O which has a 45012 gmol formula mass. MgO MgO 2 Mg 2O Only one of these is correct. Mass of magnesium oxide.
PRELAB HOMEWORK to be filled out in your bound lab notebook before you perform the experiment. Theoretical empirical formula of magnesium oxide. A compound with 0508g of copper and 0284g of chlorine.
Kindly say the empirical formula of magnesium oxide report solution is universally compatible with any devices to read 3. This is found by determining the moles of Mg and O in the product. Moles of Mg 5.
Knowing the mass of magnesium used and the mass of magnesium oxide produced you can determine the mass of oxygen used. Mass of oxygen that reacted with Mg. The Determination of the Chemical Formula of Magnesium Oxide Conclusion The purpose of this lab was to find the empirical formula of magnesium oxide.
What is the empirical formula of magnesium oxide. 3 663 words Chemistry Investigation to find the Empirical Formula of Magnesium Oxide Pages. In this lab the student burned magnesium in the form of a ribbon which bonded with oxygen to form.
Up to 24 cash back Before coming to lab write the set-ups including all units and labels of the calculations you will be making. However based on the valency we can conclude that the net charge in the molecules is zero and balanced with one magnesium and one oxygen atom. For glucose the empirical formula is CH 2O.
Magnesium oxide is formed up of the ions Mg 2 and O 2- which are joined by an ionic connection. Use the following molar masses. You will have recorded the mass of magnesium used as a reactant and the mass of the magnesium oxide product but how can you know the moles of.
A separate experiment gives a compound molecular mass of 9012 gmol. The empirical formula is HO. Up to 24 cash back while the molecular formula for hydrogen peroxide is H 2 O 2 the simplest whole-number ratio of hydrogen and oxygen atoms can be expressed as HO.
Mg s O2 g MgxOy. Experimental Determination of Empirical Formula of Magnesium Oxide - DATA COLLECTIONEmpirical Formula of Magnesium Oxide Post-Lab Lab. Although there are an infinite number of combinations We will assume that the likely combinations are small numbers and that there are three possible combinations of magnesium and oxygen that could combine to form magnesium oxide.
A sample of magnesium is weighed and burned in air. We had to find the experimental percent composition by mass of MgO come up with the experimental empirical formula of magnesium oxide and balance the chemical equation. Determining the empirical formula of magnesium oxide lab Pages.
As a result the formula for Magnesium Oxide is MgO. In this experiment you will determine the empirical formula of magnesium oxide. Empirical Formula Lab Conclusion -- Magnesium Oxide Finding the Empirical Formula for Magnesium Oxide part 2wmvEMPIRICAL FORMULA OF MAGNESIUM OXIDE - TRIAL 2 Find the Empirical Formula of Magnesium Oxide Empirical Formula of Magnesium Oxide Postlab Analysis Empirical formula of Magnesium oxide Magnesium Oxide Chemistry Unit 2.
Up to 24 cash back The empirical formula of magnesium oxide Mg xO y is written as the lowest whole-number ratio between the moles of Mg used and moles of O consumed. Divide each value by the smaller number. Why are the crucible and lid heated at the beginning of the experiment before being weighed.
Place the lid of the slightly askew. Mg02 g0224310008000812 O02 g0216001250008156 3 Thus the empirical formula of Magnesium Oxide is Mg O. Experimental empirical formula of.
2Mg O2 2MgO Moles of MgO. Therefore the molecular formula must be twice the empirical formula or C 4 H 10 O 2. Up to 24 cash back joined together to form a molecule of magnesium oxide.
The experiment is straightforward. A compound with 221g of iron and 0950g of oxygen. Oxygen 15999 gmole 1.
The empirical formula of a compound is the simplest formula based on the number of different atoms in that compound.

Lab Report Determine The Empirical Formula Of Magnesium Oxide By Reacting A Known Mass Of Magnesium With Oxygen International Baccalaureate Chemistry Marked By Teachers Com

Empirical Formula Of Magnesium Oxide International Baccalaureate Chemistry Marked By Teachers Com

Empirical Formula Of Magnesium Oxide Post Lab Youtube

Mgo Lab Report Empirical Form Of Magnesium Oxide Abstract This Lab Was All About Finding The Empirical Formula Of Magnesium Oxide The Empirical Course Hero

Name Date Empirical Formula Of Magnesium Oxide

Solved Submit Lab Report No 6 Empirical Formula Of Chegg Com

Solved 亿 Si Empirical Formula Of Magnesium Oxide Compound Chegg Com

Empirical Formula Of Magnesium Oxide Lab Report 3 30 15 Docx Alexis Sharkey Ms Joseph Chem 116 002 29 March 2015 Empirical Form Of Magnesium Oxide Course Hero

Empirical Formula Of Magnesium Oxide Post Lab Youtube

Exemplar Conclusion And Evaluation Experiment Finding The

Experiment The Empirical Formula Of Magnesium Oxide International Baccalaureate Chemistry Marked By Teachers Com
Empirical Formulas Teaching Resources Teachers Pay Teachers

Lab 10 Determining The Empirical Formula Of Magnesium Oxide Chem 100 Studocu

Solved Received 12 15 18 Formula Of Magnesium Oxide Show Chegg Com

Pdf Determining The Empirical Formula Of Magnesium Oxide Natalia Ramirez Academia Edu

Magnesium Oxide Lab Dr Kilzer S Classes
Determining The Empirical Formula Of Magnesium Oxide Lab International Baccalaureate Chemistry Marked By Teachers Com


Comments
Post a Comment